PI information
This page contains information for principal investigators about the electronic delegation log within the BSRBR-RA database.
Approving access to the BSRBR-RA database for new research team members
As Principal Investigator for your centre, you are required to log in to the BSRBR-RA database to electronically authorise a request from a new Research Team Member. The steps below should be followed in order to do this:
Step 1
An email will be sent to you with details of the researcher requesting access. This is a prompt to log in to the database. For information on Two-factor authentication, please click here.
Step 2
Once logged in to the database, follow the on-screen link to Electronic Delegation Log. This is towards the bottom of the page.
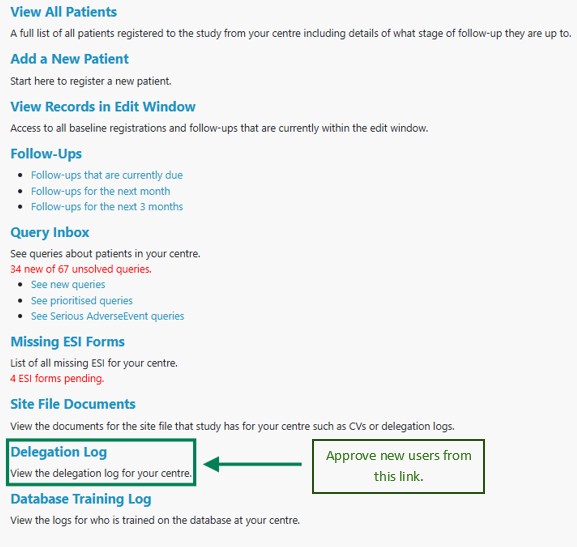
Step 3
Look for the entries marked Not Yet Approved, as shown in the screenshot below.

Click Edit to view and amend details of the new researcher's access request. Check that the delegation details (e.g. obtaining consent, performing clinical examination) are appropriate. To approve the user, use the Access Status drop-down menu to select Approved and click Update Entry.
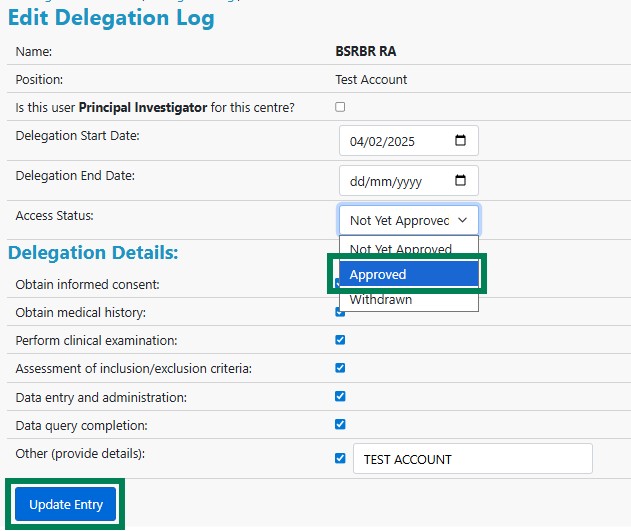
This completes the process.
If you have any queries regarding this then please don't hesitate to contact the BSRBR-RA offices on 0161 275 1652 or biologics.register@manchester.ac.uk.
Recording an end date for researchers no longer involved in the BSRBR-RA
The BSRBR-RA study team will record an end date when they are informed that an individual researcher is no longer working with the study. Periodically, we will send you an email with a list of the current active researchers at your centre. Please review this and advise us of any researchers who have left the study, so we can record an end date for these researchers.
